The Little Big History of Alkaline Batteries
How do Batteries Produce Electricity?
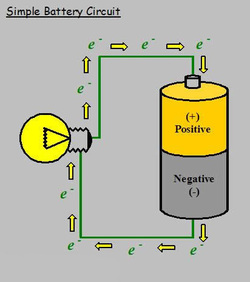
- Electricity is the flow of electrons through a path. This path is called a circuit. Batteries have 3 major parts: the cathode (+), anode (-), and the electrolyte.
- A chemical reaction takes place, which causes a build-up of electrons in the anode. Alkaline batteries depend on the reaction between zinc and manganese oxide.
- To balance both the cathode and the anode, the electrons want to go to a place with fewer electrons. The only place to go is the cathode. This doesn't happen because the electrolyte keeps the electrons from going straight to the cathode.
- When the flow of electrons or the circuit is closed, a wire connects the anode and cathode. Then, the electrons have a path to the cathode. If there is light bulb between the wire, the light bulb works because the electrons are going through the light bulb.
- This eventually stops the production of electrons. This means that there is limited power in the batteries. However, when you recharge a battery, the direction of the flow of electrons reverses.
- The electrolyte, which controls the flow of electrons, is called potassium hydroxide. Potassium hydroxide is an alkaline substance. This is why we call these batteries Alkaline batteries.
Image: http://3.bp.blogspot.com/_XMxhNbxXSkg/TPRY_4U-_kI/AAAAAAAAACQ/cM4k0Zi5Fq0/s1600/battery_circuit.jpg
